
You can contact us by phone or you can visit us at our headquarters where we will be happy to provide you with complete information, to be able to answer all your questions and curiosities about this field.
Euromedics Cryo/Bank staff is at your disposal, informing you directly and correctly.
With a single phone call to the company, we will answer all your questions about the company’s services.
Also – you can schedule a visit/tour in our headquarters to discover real-time information about equipment, process and staff.
When you contact a EUROMEDICS CRYO/BANK representative or visit us, we recommend that you have a list of questions/concerns with you.
During the briefing you will receive a complete file with all the company’s forms.
Information about the whole process
Sampling, Processing, Storage and issuance of the Certificate.
Call EUROMEDICS CRYO/BANK if you have any questions.

Collection and Transfer of the KIT
- The sampling kit that you will receive from EUROMEDICS CRYO/BANK must be kept in a cool and shady place, at room temperature, before the baby is born.
- Inform the maternity hospital of your choice in advance regarding the decision to continue the umbilical cord blood sampling process with EUROMEDICS CRYO/BANK.
- Carefully study and complete the documents that must accompany the harvesting kit.
- Leave only the child’s data blank to be completed on the day of birth.
- Make sure you take the collection kit and the file you received from EUROMEDICS CRYO/BANK with you to the maternity hospital.
- When you arrive at the maternity hospital, on the day of your birth, you hand over the sampling kit to the gynecologist or midwife. You keep the file.
- Umbilical cord blood collection can be done at any public or private maternity hospital.
- It is not important if the birth was natural or by caesarean section.
- The gynecologist/midwife does the sampling himself.
- The procedure takes only 3-5 minutes and is performed in absolute safety conditions for the newborn and the mother.
The samples are brought to the laboratory in the shortest possible time and in complete safety. The transport of the sample does not involve additional costs for the parents.
Our company guarantees the receipt of the kit from the maternity ward within a maximum of 6 hours and the processing of the sample within a maximum of 48 hours after collection.
The sampling is done as follows:
- Immediately after the birth of the baby, the gynecologist cuts the umbilical cord and gives the newborn to the nurse.
- Apoi, dupa ce curata si dezinfecteaza zona, extrage din cordonul ombilical, într-o punga de colectare care se gaseste in kit-ul de prelevare, furnizat de EUROMEDICS CRYO/BANK.
- After collecting the blood, the medical staff thoroughly homogenizes the blood with the anticoagulant from the sampling bag, using light movements.
- After the completion of the sampling of umbilical cord blood, cord fragments (depending on the package chosen) and the collection of the mother’s blood for additional tests, the Kit will be kept by the medical staff at room temperature, until it is picked up by the courier qualified Euromedics CRYO/BANK, to be transported to the specially equipped laboratory of EUROMEDICS CRYO/BANK.
Immediately after the birth of the baby, the parents will have to inform the company EUROMEDICS CRYO/BANK by calling 0800 800 855 (free call).
From this moment, EUROMEDICS CRYO/BANK will continue all actions, so that this sampling kit is urgently transferred to the company’s laboratory.
The sampling kit that will contain: the bag with the umbilical cord blood of the newborn; the vials with the cord fragments (depending on the package chosen); the vials with the mother’s blood samples will be picked up on the same day, within a few hours at most, by our courier authorized for the transport of biological samples.
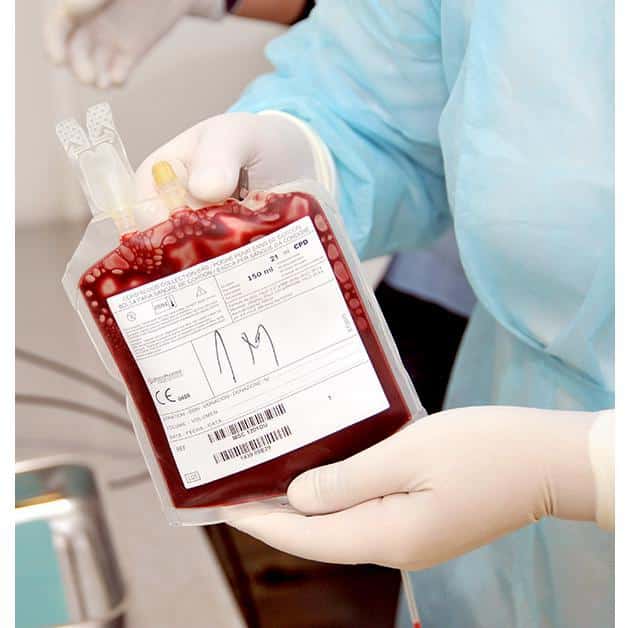
Processing of the sample taken
The bag with the collected umbilical cord blood is brought to the Euromedics CRYO/BANK laboratory, where it is taken over by our specialized staff, so that the valuable stem cells are isolated and frozen in the shortest possible time.
Immediately after receiving the sample, the data of the mother and the newborn are recorded in a special database. Each sample receives a unique identification code (bar code) and thus the code can be read by a special laser device, with the help of which we can check the data of the sample.
The quality control follows, which includes measuring the number of cells, serological and microbiological control of the sample.
Checking the volume and counting the cells is done with the help of a hematology analyzer, as well as by flow cytometry.
Next, the umbilical cord blood is separated into three components: plasma, erythrocytes and stem cells together with mononuclear cells.
The separation of the blood from the umbilical cord is done in a density gradient. Blood separation (volume reduction) is very important because only total nucleated cells are kept and the amount of DMSO cryoprotectant that is added is very small (DMSO is a toxic substance for cells in large quantities).
Immunophenotyping is performed by flow cytometry and the number of CD45+ cells, the number of CD34+ cells (stem cells) and the viability of these cells are determined. The number of CD34+ is very important for the transplant center because it represents the total number of hematopoietic stem cells in the respective sample (CD34+ is specific only to hematopoietic stem cells). The sample validated for freezing (viability over 70%) is transferred to a special bag (cryogenic bag) and then subjected to gradual freezing. Cell viability is realized in an automatic system by flow cytometry.
After it has been gradually frozen (using the Ice Cube 14 S instrument), the sample is kept in quarantine until the final validation according to the results of the serological and microbiological tests.
The sample is stored in a tank with nitrogen vapors and in metal boxes, which both allow for storage for more than 20 years, but also prevent contamination of the samples.
The verification of the sterility of the umbilical cord blood is carried out by blood culture in the Bactalert automatic system.
The umbilical cord tissue is processed by two methods: processed tissue stored for eventual extraction of mesenchymal stem cells and extraction of mesenchymal cells from Wharton’s gelatin.
The extraction of mesenchymal cells from Wharton’s gelatin is carried out by an enzymatic method and the processed tissue in its integral version is stored for subsequent cultures.
In accordance with the competent world organizations and EU requirements, the following tests must be done from the mother’s blood, during the 9th month of pregnancy or at the latest on the day of birth, as well as 4-6 months after birth:
- Cytomegalovirus
- HIV I & II
- HTLV I & II
- Testing for syphilis is also done.

Storage of the sample taken
Our laboratory is located in a specially designed space, in the new headquarters – an investment made by Euromedics in Bragadiru, Str. Unirii 83A, very close to Bucharest and equipped with the latest equipment.
The storage room is specially arranged, ventilated and video monitored 24/24.
The temperature and humidity in the room, as well as the temperatures of the storage tanks are monitored non-stop, with online access for Euromedics laboratory staff.
The cryogenesis of stem cells from blood and umbilical cord tissue is done in a progressive and controlled manner, after the addition of a cryoprotectant substance, thanks to which the stem cells are protected and remain viable for decades in storage tanks with liquid nitrogen vapors, at very low temperatures , close to -196° C.
The entire procedure is recorded in a computer program, thus facilitating the finding of each sample stored in the Euromedics laboratory.
The final bag of blood containing the stem cells isolated from the umbilical cord blood is placed in a special protective metal box and together with the sterile cryotubes containing the stem cells from the tissue (Warton gelatin) are inserted into a state-of-the-art device, of progressive freezing, where, by means of the computer, the temperature is gradually and precisely lowered to -100° C, so that the integrity and viability of the cells are not endangered and then introduced into the storage tanks with liquid nitrogen vapors, where they can be kept for decades.
In each storage tank there are labeled metal supports for cassettes and cryotubes, so that each sample is placed in a certain place, with its own identification number.
Issuance of storage certificate
The entire laboratory procedure and sample verification is carried out within a week.
All the data about the collected umbilical cord blood and umbilical cord tissue, its unique code, the number of hematopoietic and mesenchymal stem cells, as well as all the results of the laboratory tests performed, are presented in a final certificate, which is given to the parents.
The certificate will be useful to the parents in case of a transplant, which will eventually be performed on the recommendation of a treating doctor.
The following are specified on the storage certificate:
- the total number of CD45+ nucleated cells
- the total number of CD34+ hematopoietic stem cells
- the total number of mesenchymal stem cells
- cell viability expressed in percentages
- the result of the microbiological testing of the umbilical cord blood

